Hi All,
As of late, it seems like every 3rd post on the forum has been about the filtration of various components from EtOH crude–so I’m gonna hop on that bandwagon.
Some of the components that require filtration are, in no order:
Hydrophobic Molecules - Epicuticular waxes, lipids, aliphatic hydrocarbons, generally things that can be precipitated from ethanol crude by lowering temperature.
Polar Undesirables - Chlorophylls, Flavonoids, etc. Chlorophyll, in its many forms, is pretty polar. Polar molecules are highly water soluble.
Particulates - Insoluble matter, usually remnants of biomas that have made it through the initial bag used during extraction.
In my opinion, it is helpful to consider at the size of the molecules as a starting point for filtration choice.
This is a picture of some crystallized epicuticular waxes from various grasses–let’s assume that crystallization has occured to a large extent in pre-winterized, cold, EtOH crude. If this is the case we’d want a filter size that excludes molecules that are (at most) the size of these crystals. Given the exact scale here, it looks like these waxes are a diameter of about 1-5 microns.
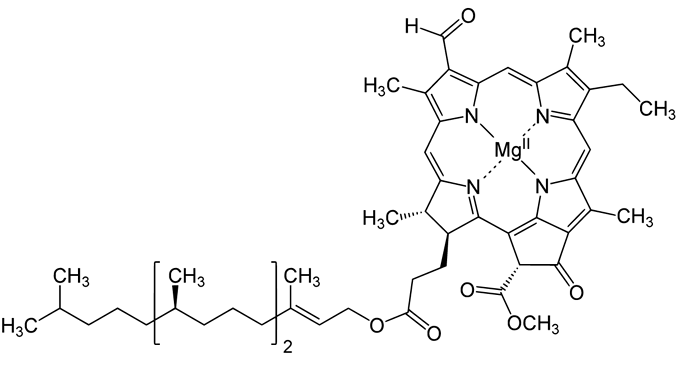
This is a picture of a generalized Chlorophyll molecule–its pretty large relative to a molecule like water, but too small to filter using (non-membrane) size exclusion filters (micron bags, cartridges, etc). So, afaik, these molecules must be separated by manipulating their polar character. For size, i’m going to call these too-small-to-filter-with-size-exclusion.
Particulates don’t really require a picture, these are solid particles that are often visible to the eye, but also slightly below the threshold of visual perception (< 1mm). A safe bet is to assume that these molecules are in the range of 1micron to 5mm.
So in order of size: Particulate > Waxes > Polar Molecules.
In my experience, process fluids that are to be filtered several times will be filtered in order of decreasing particle size. So the above ranking would also serve as a basis for the design of the filtration train.
So, the thing that folks mainly care about is what type of filter to use:
Bag Filter - Available from a fraction of a micron to several hundred micron. Usually constructed of a polymer (polypropylene, PTFE, nylon, etc).
Cartridge Filter - Also available in various mesh-sizes, offers a much higher surface area for filtration. Cartridges can be packed with adsorbents, like activated carbon, silica, bleaching clays.
Lenticular Filter - Also available in various mesh-sizes. High surface area, can be packed with adsorbents. These are stacked discs of filtration media that are easily backflushed and should be easily reusable with appropriate maintenance.
(Note: Lenticular filters are a type of cartridge filter, I just wanted to separate for the sake of comparison. Cartridge filter here is meant to imply a non-lenticular, single-column type filter.)
Plate Filter - A plate filter uses hydraulic pressure to pass the process fluid through a series of filter sheets. Various mesh-sizes available. Not sure about adsorbents?
Buchner-Type Filtration - Buchner filtration can be done with various mesh-size filter paper, adsorbents can be added in desired quantities. This is easily the most customizable solution, but often times the slowest.
The Primary Filtration step, to remove particulates, is generally the easiest–so it should require the easiest, cheapest, solution. I say this, because particulates can be filtered by decanting–so there’s no need to buy the most expensive unit possible, just to grab the pesky f***ers. In my opinion, this step should be done by heating the solution so that its viscosity is as low as possible, and pumping it through a reasonable mesh size (10 micron? 25 micron? 100 micron?). A bag filter is totally sufficient for this purpose, but so is a cartridge filter. I don’t have enough qualitative data to make a suggestion on size, but I can say that I would choose the option with the better throughput. Determine which system has a lower pressure-drop (probably bag) and go from there. My recommendation is a 25 micron filter bag with a good pump.
A pig filter might be a good choice here, because the hydraulic pump could be used to modulate flow to any desired rate (as long as the solution has been warmed up sufficiently to solubilize waxes).
Winterization poses some interesting problems. 1) The solution is very cold–use a good stainless steel housing. 2) As the solution filters, the filter itself becomes quickly fouled–the waxes begin to accumulate on the surface–it wouldn’t be a good idea to use an expensive filter here. 3) As waxes accumulate, the mesh-size is effectively decreased, the waxes are acting as an additional filtration layer.
It seems to me that the most important factor in choosing a correct winterization setup is which system is capable of the highest throughput. I’m drawing a complete blank here, but it seems that the most desireable option is a bag filter. I have a feeling that cartridge filters will foul up too fast and the increased pressure drop will slow the process dramatically. Either way, a good pump is required. I would certainly not recommend a pig-filter, because a huge portion of waxes will collect on the first plate–i.e. the pressure drop increase is not distributed amongst all plates, it is collected in the very first one–the process is slowed from the very entry point.
Final Filtration (Polishing) - In this arena, the clear winner is the lenticular. The extract is so much cleaner by the time it arrives at this stage (if the previous filtration steps were adequately taken care of), that there should be relatively little worry about pressure-drop changes over the lifetime a single batch process. The filters are designed for back flush, so you are capable of cleaning after each run. With appropriate maintenance, there should be no concern of excessive replacement costs. Typical adsorbents for color remediation would be Activated Carbon and Bleaching Clay adsorbents.
Pumps - I’m not too versed yet on the appropriate pressure generation unit to use for this application, but I like the idea of using a double diaphragm pump because of its safety and the relative simplicity of the design.
Conclusion: If I were to design my perfect system — 1) Filter warm crude through a plate filter. 2) Chill crude to -50 and filter through a bag filter. 3) Warm crude and filter through a lenticular housing with activated carbon. 4) Maybe getting a little crazy here, filter through additional lenticular housing with bleaching earth.
Feel like that’d make a pretty good feed to a distillation unit.
All critiques, additional information, general discussion, and kindness are appreciated.
-Phil