It improves clarity big time
I was under the impression that you were doing one with wannabe in Eugene
Well that’s false and if I do a class in the future it would be at my lab in San Diego where I would have all the tools I would need
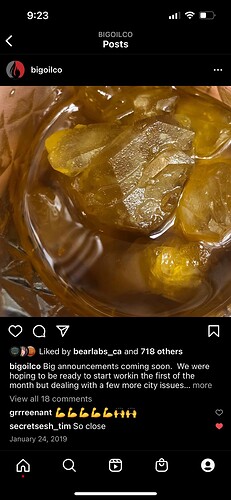
Looks are deceiving with fast crash. This was nowhere near only thca and tested at 8% terpenes. this was later remelted In the oven and was a wet badder.
The fast crash happens at a rate that is hard to imagine and locks in all the terpene and minors throughout the whole pan.
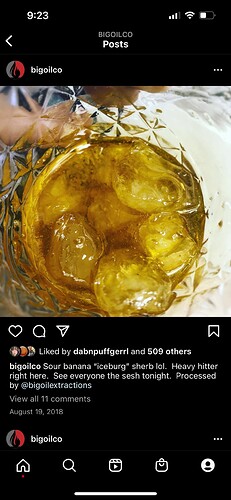
Here a picture of that same pan after breaking it up before going under heat, you can kind of tell in the photo how sticky it is and clumpy and how’s it not like a regular cold crash where it’s thca is like a dry sand.
All good. I wasn’t aware that the plans changed.
If it’s 90%+ it’s going to cold crash fairly easy. Doesn’t necessarily have to be isolate to be considered good gas when it cold crashes.
Now if it’s an impurity that forces thca out of solution, then it should do it with anything with thca in it right?
Once the “clean gas” supplier comes, I would like to try the Medusa SOP with it. That should answer it for sure
Fairly easy? Or aggressively fast without giving it any favorable conditions to crash?
Can you crash out an ENTIRE pan from cured trim fresh out of the machine in less than 10 minutes leaving no solvent behind?
I don’t see typical cold crashes boiling off all the solvent because at some point the solution drops out enough thca and solvent is left behind.
You said yourself you haven’t seen it happen without using isolate, and the pictures you shared look like a standard cold crash and not a bad gas fast crash. The contaminated fast crash gas will pretty much always boil away all of the solvent long after the solution should have stabilized.
I’ll try and get a thermometer into a fast crashing pan and I’d bet it’s getting below butanes boiling point. I say this because I have had ice form on the bottom of the pan before while it was still actively boiling solvent away.
Kinda like the boiling point of its redheaded step cousin
Those jars were capped which is why there is still solvent left
And do you think they would have fully evaporated at a room temp of 60-65f in less than 10 minutes from being poured?
Indeed. You can’t say you know fast crash unless you can pull video of the above. This is rapidly becoming a circlejerk, apart from those that have seen the WTFamine in action.
It’s not even good bio doing this. Years ago, you’d struggle to get bad bio to crystallize out, and now you can sugar it in minutes? And we’re suggesting the butane is cleaner, not dirtier, despite the very well documented issues on all flavors of the gas supply chain? I call bullshit…
Frankly, I’m just grateful it’s not happening in the collection vessel. I’ve seen examples of that in person, and I would hate to wrangle that fucking clog.
Below are pictures from 3-4 years ago some with media some without. Never had any of these issues growing these stones at 60-75 degrees. Hell my lab in the winter would get down in the 40s.
How are we so sure it’s an impurity, yet we have no solid data yet? If it coats the thca crystals all the way through, every thca molecule having one impurity on it to chalk it, then why can’t we test for it. It would have to be in large amounts to be able to do that to all the thca thoroughly. It’s just butane being trapped hence why we see it show up as soon as you purge the rocks
@johnbigoilco when doing those temps in say pentane for example the diamonds won’t chalk, neither will they fast evap of course being pentane. If the old gas had any pentane or propylene or other gas, it will raise its boiling point which in turn could prevent the Medusa you wouldn’t see before and the fast crash. If the the old gas was already running at the bottom of the barrel before the switch came, why would the contaminants be in the new gas, I would guess its in the old gas. Also if the butane is gassing off easier then it would have to be purer, same if it’s crystalizing faster
I may be wrong here, but I was under the impression that rexing in pentane also removed the Medusa issue, but there’s a noticeable difference in loss when it’s rexed.
Just vomiting up what I think I remember
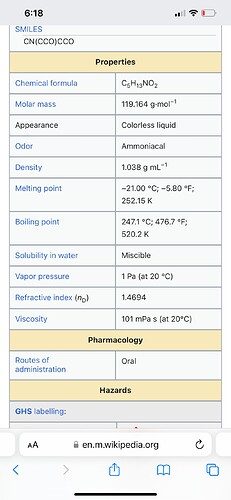
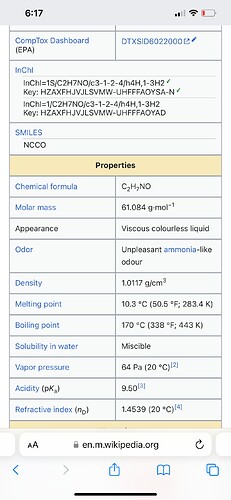
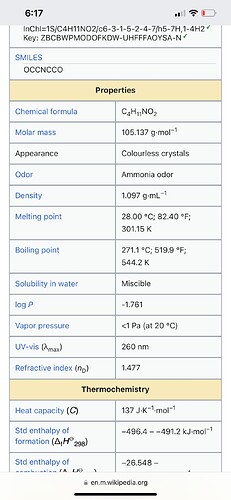
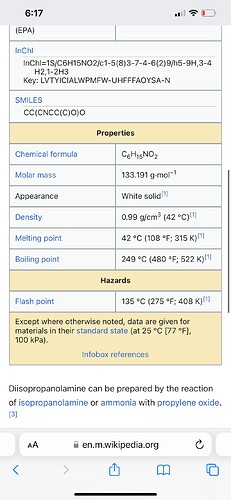
You guys think the oil and gas industry just barely started using amines? Here’s some details about the amines used. Check out their boing points, mass, and appearance
Amines are used to remove meoh from the crude source. In early 21 the main source of crude for the refineries changed from a very pure almost non sour crude to a very nasty extra sour batch. Sour means it’s full of sulfurs. The meoh removes the sulfurs and then amines are used to remove the meoh. If the starting crude needed more meoh then normal to sweeten then the amount of amines would have increased.
We have tested the gas the problem was when we tested it we didn’t know what to look for.
Remember we use less then 1% of the butane made. The refineries don’t care about us. Your gas for your car can have up to 10% butane in the winter and 2% summer blend.
My guess is we are about 2 months away from having an upstream solution. I have some info I’ve been asked not to share but gas has been found to have a few things in it that are not supposed to be there.
Also just got a new employee a few weeks ago That has been making diamonds just as long as myself I wanted to bring on somone that had a different perspective/sop on diamond making. He was of the opinion it was an sop issue (he had not seen the fast crash or Medusa) . Once he started doing runs he quickly became aware of the issue and was like “ok your right the gas is acting different”.
The part I don’t understand if it’s not the gas is why has my tried and true sop stopped working. As you can see from the pics in my last post, our diamond production was on point. Took longer then I would like to fully crash out but I was always able to get beautiful rocks. So what changed if the gas didn’t. If you say biomass I will refute that with my own material I’ve been growing outdoors the same way for the last 10 years also has the same issues now. @Dukejohnson and @Dred_pirate both have a similar story to me and had no issues with our sops working 18 months ago.
If you read @Photon_noir’s thesis, that is not the claim.
If you think the claim is the amines are newly in use (e.g. have not been used in the crude refining industry prior) , I recommend you reread the entire “It’s not Isobutane” thread.
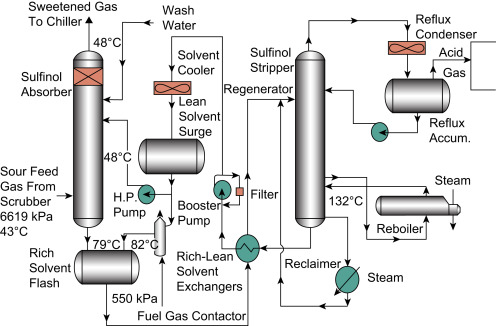
I’d also recommend reading @SafeLeaf.ca 's detailed explanation of how crude fractionation → butane happens, along with a few handy flow maps that show how sour gas is treated.
Let’s summarize
Fun flow diagram:
Amine Carryover aka Issues in the Stripper
In the Stripper, the equilibrium value for Ammonia from source feed into the Condenser is considerably < 1, causing Ammonia vaporized in the Stripper to be returned into the Reflux. The equilibrium value for Ammonia within the Stripper is a bit > 1, resulting in a mild tendency to exit the Stripper overhead rather than the bottom. So the Ammonia concentrations tend to build up in the Stripper as more Ammonia tries to exit the top, which causes it to be collected and returned into the Reflux again.
If the gas supply is contaminated which I sure looks like.
What recourse do you businesses owners have? Do you think you can sue for losses? Or are you just recrystallizing it all which seems to remediate the Medusa stones….
[quote=“johnbigoilco, post:197, topic:178019”]
Remember we use less then 1% of the butane made. The refineries don’t care about us. Your gas for your car can have up to 10% butane in the winter and 2% summer blend. [/quote]
You’re right we’re so small they don’t care about us. I suspect we had been using a blend back then with cosolvents with higher boiling points in there. The pattern I see is that if the gas was boiling slower and not getting as cold back then, it was because of the other solvents in there. If it’s pure or has say isobutane or propane in it, it will boil faster and get colder