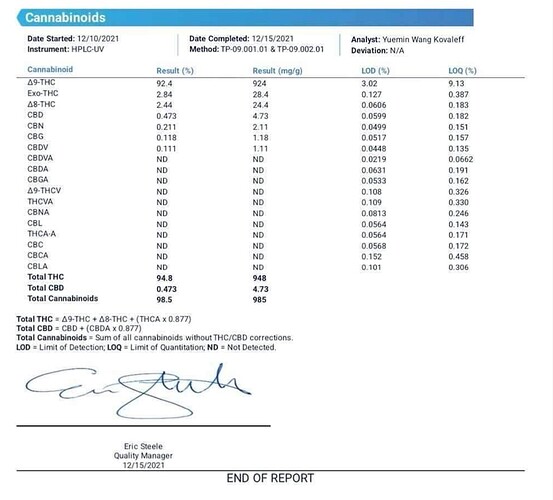
is this a converted liter ? 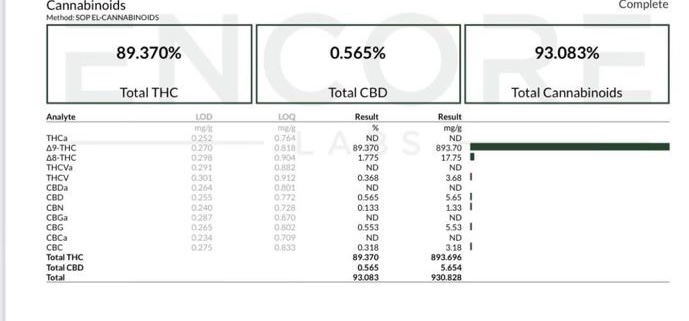
Hard to say,
If theres really cbg in there and its not a misidentified peak I’d say its from biomass
Up to 2% delta 8 is normal for distillate made from CRC material.
I haven’t seen Cbg in converted liters. The low d8 is pretty normal on d9 disty
If you can see the graph. Usually you will see a lotta unusual peaks before the cbd on a converted oil.
There’s usually a little cbdv in non-converted as well, no?
I think the iso THCs are the markers of conversions, iirc @kcalabs is working on standards
This is converted.
1.775% converted guaranteed.
Sprinkles cbg
ND d8 sop whaaaa
Probably not converted. What makes you think it is?
Could be converted they just added a small amount of mother liquor from cbd isolate back to get that full spec and more “traditional” color.
There’s a chance it could be from biomass as well that converted from left over media or not being the right ph
Is there an uncommon isomer that a lab could get the standard for to test legitimacy of distillates?
Like, if you converted, there’s gotta be imperfections at SOME scale, right?
Yeah I agree. I don’t know how they could get the ratio of D9:D8 so high.
This is from isolate
It’s called triisobutylaluminium
Exo thc or iso thc are for sure signs of isomerization
cough cough Chain of custody, seed to sale tracking, reliable vendors cough cough
Based on the color alone I’d call that a conversion
Triisobutylaluminium gives weird colors usually haven’t been able to figure out why
Color seems like converted. That peach color is something you see sometimes.
Any idea why this happens?
The post above the left 2 are heptane, the right one is dcm
So far as I’ve seen since conversions entered the market: the more dangerous the process, the better the product.
This is especially true for solvent. In terms of desirable end product it seems like:
Heptane < Hexane < Toluene < DCM
There’s a big ass book on my computer about “Solvent Effects” In chemical reactions, I’ll send it to you when I get home. There is a very real difference in the outcome of catalytic reactions between different solvents and I couldn’t tell you exactly why, just that I know it happens.
Also, I might mention, the higher the heat the worse the color.
If I had to make an educated guess I’d say that a better solvent solvates the reactant, product, and catalyst better than a worse solvent.
That better solvation will lead to more “relaxed” solvent molecules, possibly leading to reduced friction, density, viscosity. Additionally, it is known that reaction rates and reaction activation energies are directly related to solvent choice.
So if less energy is required to reach the transition state and hit the downward trend on the free energy vs time chart, and less energy is generated from friction, etc…the process doesn’t get as hot and “weird shit” doesn’t start to happen in terms of color (whether that’s entirely different molecules or just oxidized materials or quinones, who knows).
i think this one could go either way. although the TCA is a little low so unknowns could be concerning. if it is converted its a decent conversion… but globtek could be spot on with what he said