I do volumetric dosing on a 50 gallon batch tank for roughly 30 plants. I use peristaltic doser pumps, with concentrated feed. I am able to mix batches on the fly with VERY little variance as long as the concentrate is made correctly.
Using flow meters would be to mix large amounts of concentrates accurately at a larger scale. For preparing final nutrient solutions adding nutrients and then water till you reach the target EC is an acceptable way to do it. Flow meters and weighing are not meant to help you prepare final nutrient solutions, just concentrates.
@danielfp
Hey Daniel, would you have a quick look at this post if you have a few minutes (there’s only a few posts related).
A magnet easily attracks and “pulls” the iron (assuming) out of this fertilzer mix (it’s Plant-Prod 6-11-31).
I’d really like your thoughts on what this means, what possible implications are. The subject came up because a user mentioned that he thought his magnetic stirrer (and stir bar) was pulling the iron from the mixture he put on to stir.
(First post it was brought up: Jacks Nutrients - #264 by Rocket - Overgrow.com).
Anyone else want to share their thoughts on this as well?
Thank you.
Thanks for sharing. So Fe salts can be paramagnetic, often metal chelates that contain Fe3+ (like the ones used in hydroponics) can have paramagnetic behavior and be separated by magnets. However this behavior only happens in the solid state, once the fertilizer is put in water, the organized crystal domains required for this effect no longer exist and while the Fe chelate molecules themselves are paramagnetic, they are not attracted to the magnets once in solution because their orientation is random once they are dissolved.
If you put a magnet while your mixing, some solid will temporarily stick to the magnet, but will eventually all dissolve. This is nothing you should worry about. For anything to “stick” to a magnet, there must be large groups of atoms or molecules with organized orientations that can interact with the magnetic fields, no such thing happens in solutions. Only solids can do this, or solids dispersed in liquids (like ferrofluids).
Hi
Do you pse have the Hydrobuddy
Element conc PPM values for GARLIC
thx in advance
Hi there, I am a novice grower and would love assistance with trying to “adapt” the natural 310 PPM tap water to my grow. I have the most recent water breakdown statistics from the region of Waterloo, but still cannot make heads or tails of what my plan of attack is.
I am a big believer in donating, especially to open source projects - I make upwards of 50 donations per year!
And would absolutely be thrilled to donate if I could possibly get this working for me.
I’d donate to both this forum, and the software.
The thing is - I don’t use base nutrients but rather am growing in organics (Black Swallow KIS Mix) so I’m not sure it’s exactly the same. But I need to adjust my water source to make it perfectly viable for my plants, and have not been able to figure this out yet.
If additions to the water were needed, then my local soil supplier, Black Swallow Living Soils, sells ALL the amendments individually for maximum savings and flexibility in adjusting things as needed.
Huge thanks in advance for any help, and I look forward to any assistance!
Premade organic mixes won’t play very well with hard water like this. Organic mixes are normally quite Ca/Mg rich and the water will just pile on to this heavily and cause imbalances. You would need to custom formulate your own organic mix to make sure it’s low in Ca and Mg if you want to use this water source with it. This means your mineral amendments need to be quite limited (no bone meal, gypsum, etc).
You also absolutely need to lower the pH of the water with a mineral acid - sulfuric, phosphoric or nitric acid - before using it. If you are not adding more nutrients to the water then phosphoric acid is ideal. Do not use organic acids (ascorbic, citric, acetic), as these will create pH imbalances in the media due to the way microbes will metabolize them.
Seems like everything you said makes sense and is likely what I am experiencing… It’s also very useful to me that you mentioned the acids thing as I have both citric versions which I’ve been using up so far, and unopened General Hydroponics Phosphorous version which I didn’t even crack the lid on yet. Maybe a source of my headaches is due to repeated pH downing with the citric based item as well…
Is it safe to treat an entire 5 gallon bucket of water with just a pinch (0.2g) of Absorbic Acid? I treat for chlorine and chloramines by using Abosrbic Acid and dose it according to this handy Aquaponics spreadsheet: Here is a water dechlorimination calculator spreadsheet for everyone. : r/aquaponics (reddit.com) and it generally comes out for me suggesting around a mere 0.2g pinch to take care of the entire bucket.
And secondly, should I still dose the 0.2g Absorbic into the 5gal bucket if I end up switching to straight RO?
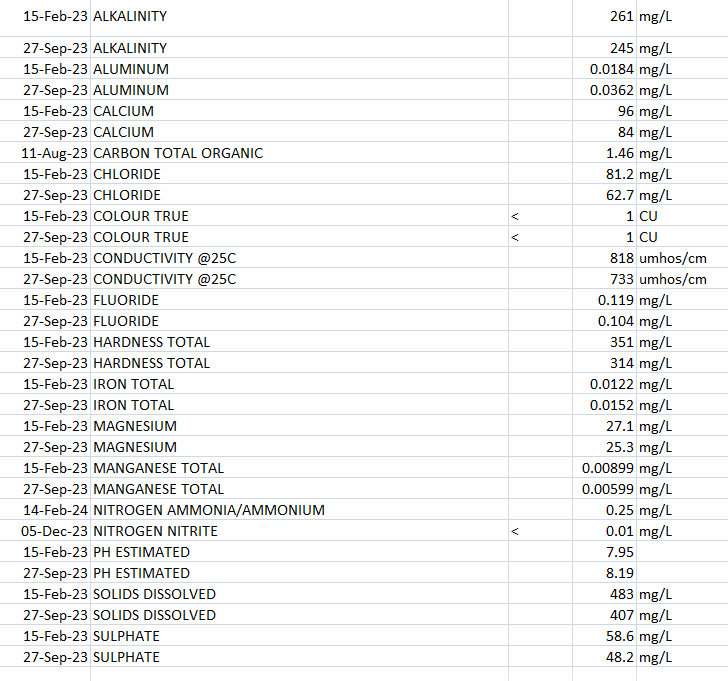
Lastly one last question, since I have you here, these are the results from Mannheim Water Treatment Plant which supplies my town with water:
Would you recommend eliminating this water completely and switching to a 100% RO regiment? Or would I be able to incorporate any of this tap water into a blend? Would it be useful or not? And if so, what PPM would you recommend taking it down to? I was going to experiment with diluting down from current 310 out of the tap to 150. But I can get it down to 10-15 PPM if I use my RO dispenser.
Huge thanks again for any help!\
Also, your post mentioned hard water and supersoil organic mixes = bad news. I also have ProMix HP here which is inert, would this be a better choice? It’s much easier to water too, holy!
Edit: Ahh, I see support from you is paid and not free, there is a big disclaimer on the HydroBuddy website that support needs to be booked and paid for which is totally understandable. I appreciate your time and help regardless and won’t expect anything else for free
The very small amount of ascorbic acid would be fine, that is just to react with chlorine, not to significantly acidify the water. You do want to stay away from any organic acid for acidification, as I mentioned above.
If you want to grow organic using your tap water, you’ll just need to blend your own soil. Same thing if you want to do pure hydro, you’ll need to blend your own nutrients. You can certainly go to RO water and just use the standard things.
I can certainly help you with the design through consulting. Feel free to book an hour through my website if this interests you.
Thanks for the information here, Daniel.
Just an FYI, I’m getting a “critical website error” when I try to open a blog post in pdf format from your site.
Thanks for the heads up, I’ve fixed the problem now.
No problem. Thank you.
Question regarding your blog post on nutrient availability and pH (charts). It ends with with:
“I believe we can create a chart that is specific to nutrient availability in hydroponics, perhaps even charts that show availability profiles as a function of different media. We have a lot of experimental data on the subject, product of research during almost a century, so I believe I will raise up to the challenge and give it my best shot. Together, we can create a great evidence-based chart that reflects a much more current understanding of nutrient availability as a function of pH.”
Has this happened? Is anybody doing this?
@danielfp Another question for you.
I just got some calcium chloride to try out. Usually, I mix in a beaker with mag. stirrer, in about 900L of water (approx. depending); ~100g plant prod 6-11-31, ~30g MgSO4, and it mixes perfectly clear (green) within several minutes. I just tried to add the calcium chloride to this same beaker at ~11.5g. The results were…cloudy is the best way I can put it. And when turning off the stirrer, it almost separated a little bit - some of it looked as it normally would and some was sort of cloudy.
I poured it into the res. of about 22L of water with a mixing pump in it. When I got to the end of pouring out the beaker there was white kind of sludgy residue.
What did I do wrong? Can I not mix the CaCl in with the PP 6-11-31, or just not in that conectrated amount (in a beaker with only ~1L of water)?
The product I used is “Briner’s Choice Anhydrous Calcium Chloride Pellets”:
https://www.oxycalciumchloride.com/products/briners-choice-anhydrous-calcium-chloride-pellets
https://sds.oxy.com/DirectDocumentDownloader/Document?prd=M48001~~PDF~~MTR~~ANSI~~EN
I did read this on the info. sheet, but ended up buying some to try it anyways:
“…This product is intended for use in solution form. After dissolution in potable water and filtering or settling to remove insoluble residue…”
You cannot mix Calcium chloride with a 6-11-31, Mg sulfate or any other sulfate containing product in a concentrated form. When you mix it you will form calcium sulfate, which will precipitate out of solution (the sludge you observed).
You can however add both the 6-11-31+Mg Sulfate and CaCl2 to your reservoir independently, to reach the final dilution amounts.
Thank you.
I tried mixing the calcium chloride in the beaker I mix the calcium nitrate in and that seems to work fine as well…
Yes, calcium chloride is compatible with calcium nitrate, you can mix them together. Just keep calcium chloride away from any concentrated sulfate containing solutions.
Hi , which is the reason.
Thanks
I am able to open the program in Mac (M3 Sequoia) but unable to enter data into the fields. Is this something you are able to advise on @Pussybandit?
Hi I am studying horticulture and would like to know what calculations are typically done in hydro Buddy to achieve the results.
I’ve read about formulas for calculating the desired amount of N from a fertilizer then figuring out how to calculate the other elements based on the % of N desired.
I’ve downloaded hydrobuddy but it doesn’t work on my Mac m1. Since it is open source is their a way to see how the behind calculations are carried out ? Does anyone have a link to a website that might show how to do these. The handbook of chemistry and physics is about 3000 pages so hopefully something a bit more accessible to begin with.
Been a long time since I came here. I just wanna say we love you @danielfp. Hope you’re doing great, man.