You do not need heat or pressure but you can give it a go if youd like. My suggestion would be toggling with positive pressure. This is termed as a method for forced crystallization and is used widely in chemical processing. However please be careful putting volatile solvents under positive pressure, this is also referred to dramatically as a bomb…
This seems almost on topic: ran across the following statements that I’m hoping the brain trust here can either confirm or deny.
When using ethanol as the extraction solvent, the temperature can somewhat determine whether THC or CBD are extracted more heavily.
Supercritical CO2 is the most customizable method, allowing for individualized extraction methods. At a specific temperature and pressure—around 80°F and 1100 PSI4—CBD is the only compound that will be drawn away from the plant material by the CO2solvent.
Edit: on the fence about providing the attribution. pretty sure y’all can figure it out if you care.
I don’t think the statement about ethanol is true at all. Ethanol extracts more CBD than non-polar solvents, and it is always going to prefer CBD, no matter the temperature, but it still extracts THC just fine.
CO2 is customizable, but it is always non-polar and it’s actually a rather poor solvent for cannabinoid acids in general. That temperature and pressure would extract THC and CBD based on the presence of terpenes acting as cosolvents. So that, too, is false.
Will this process work with dewaxed crude testing above 60%, or have you found distillation to be a necessary step?
Hey hey, I’m working with several farmers growing cbd here in western Kentucky, I am also in possession of two large stainless stills was hoping to start up a lab in the the next6 months. Can you help?
Yes, message me
Hey guys, had a small question.
From time to time when I isolate CBD from distillate, the whole solution will sugar up (pictures below).
No proper seperation. What is it due to?
Is it because the cooling is going too fast and isn’t allowing proper seperation?
!Not fully dewaxed?
try more solvent, looks like you formed small crystals.
I already tried adding more solvent and the same things happens. I honestly think it’s because I’m cooling it too fast.
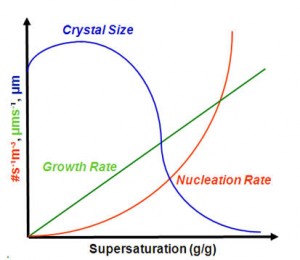
The less super saturation, the larger the crystal. Add more solvent. Don’t cool so fast if you’re worried. Take your time. Big crystals take a lil longer unless you’ve really honed your process.
I just wanted to add: if we look at the mixture you’re attempting crystallize, we have to consider that all the components have different solubilities in your solvent, solubilities which are all dependent on heat in different ways.
It might simply be a matter of: too many different components crowding up the mixture and therefore several different crystal structures forming alongside each other?
@Photon_noir is the resident chemistry whiz as far as I can tell, maybe this question interests you?
You have a rough plot, Man! How can I possibly improve upon that? Certainly with all the inherent variables, I cannot. You are correct. Nothing to add, here! ![]()
What’s up man, I’m hoping to get some tips on crystallizing CBD using some eTOH extracted crude and pentane.
I understand the basic concept of it all but have some questions still. I am without a chromatagraph but understand it can still be done. I’d love to talk to someone who understands the process.
I’m in the process of designing a semi-large scale setup for making CBD isolate. I’m currently working with hemp materials (as the flower material is still not legalized in the state I’ll be working in). I’m really working around the 0.3% THC factor when considering my design, as the product would be marketed under CBD licenses.
I’ve proposed several solvents to consider when performing the extraction, but I am personally not well versed in knowing which will yield the best results for short path systems. For instance, I’ve recently read good things about EtOH in short path, but I know the extract can be a headache to deal with considering all the BS you pull.
Would you be able to details some of your industrial setup and/or comment the use of several solvents. I’m mainly considering designs with pentanes, aromatics, and simple alcohols. What are some obstacles I should consider when designing?
@TheDarkStallion Cold EtOH doesn’t seem to have that issue with pulling all the BS. Our current CBD supplier sold all their CO2 equipment recently in favor of ethanol just because they’ve been able to process so much more material. Looks like the key to clean crude is keeping it cold, though.
Tried sending you a PM about your isolate process but I’m not allowed to. Do I need to be on here more than 5min to do so?
I’m trying to run some preliminary lab experiments to isolate CBD in a large column. What solvents are generally used to do this? I have plenty of hexanes, DCM, and AcO, but I can’t find any good papers on which solvents work best and if I have to process the eluent any further after the first run.
Also, is there a better system to learn about rather than forward phase column chromatography? I’m lab trained, but I don’t have any experience with fancy reverse phase, and orthogonal flash chromatography. Thanks all!
IT doesn t ![]()

