![]()
So you’re saying @beaker and @circus_animal had it right (or damn near)?!?
![]()
So you’re saying @beaker and @circus_animal had it right (or damn near)?!?
I totally forgot about that part!
I read all the procedures mentioning to pH balance otherwise the distillate would turn purple. Knowing that and religiously pH balanced this before solvent recovery. The first batch went purple. I pH balanced it again, same problem. Then I said fuck it and balanced the pH to 5 and it still turned purple. That’s what made me investigate magsil more. Thanks for bringing that up, that was an important detail
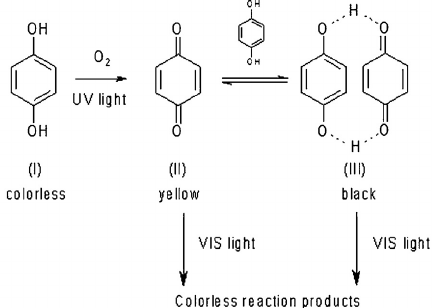
I actually made a crude mechanism for this and I think the oxidation reaction consumes oxygen gives off water and the reverse reaction does the opposite. I remember the purple color disappearing during distillation. If anyone has an FTIR, I bet a water formation peak would show up, though there may be a lot of noise compared to the signal since these colors are so strong the quinone could be in ppm levels.
I believe the vivid colors likely come from phenol-quinone coupling. The quinone may be less stable upon time, and further degrades to a yellowish compound under action of O2 and/or UVs.
I know this thread is old but if anyone is looking for the perfect solution to prevening HD9 liters from turning purple, DM me! Happy to connect
hey guys so yesterday I was experimenting with water washing a solution of pentane + thc in sodium carbonate water.
After washing it, the color of the water stayed perfectly clear.
But now this morning the water has turned purple. however, the thc stayed perfectly yellow
So my guess is that the purple impurities are the degradation of water-soluble non-purple impurities in the thc (I made mine with cbd+zeolite)
Search button is your friend. Use it.
Ya basic
(Alkaline)
I’ve actually found that with d9 acidic pH can make it a little bit more maroon.
I’ve had to pH shift some products a few times because you could distill it a thousand times and it would still come out red even though it was water clear while it was distilling.
And all that was is a pH shift to slightly basic honestly for neutral d9 but for acidic d9 you want it to be shifted slightly acidic.
To do the pH shift you need to put the product in something that is slightly polar like ethanol or acetone so that way the acid or base can actually affect the molecules pH.
Correct its pH and then filter and recover
THCA / CBDA / whatever-A will react with bases to form salts that are water soluble and typically a maroon or purple color depending on the cannabinoid. Like CBDA makes a nice rich purple while THCA makes a dark maroon.
This is one of the biggest problems with CRC is if you don’t pH your media and you’re slightly basic you or THCA is going to get basically pulled out of your extract you’re going to have higher terpene content going to lose some of your yield and your terpenes are going to taste kind of funny. Because they will react with acidic and basic media and isomerase and are not required to have it hot like you would see with Delta 8.
How are you balancing ph inline? Do you add powders to water til it reaches 7 and track the ratiis?
What I found to work best is I will pH all of my media first so I’ll put a little bit in some distilled water and then hit it with a pH probe and see where it lands at then write that number on the bottle and then whatever percentage of that media I’m using I’ll multiply by that pH number and in the end my blend will usually land right where I want it to be at nice and neutral
You’re adding multiple percentages to get your final percentage.
If you balance your medias properly you’ll have a very nice and neutral output.
And then at that point the only thing is to do is not get too hot or cold while you’re running through it and make sure you flush it nice and heavy because terpene alcohols travel much much slower through polar media than hydrocarbon terpenes because of the alcohol OH bond makes them bond through the polar media and now they stick to it and they don’t travel so fast through it and that’s where you end up losing terpenes
This effect is even more pronounced with D8–I once distilled some poorly neutralized D8 and it turned bright magenta.
I’ve seen all colors of the rainbow come out of Delta 8.
It can be green it can be pink magenta orange purple blue… you name it I’ve seen it.
I have some thca isolate I decarbed that was not winterized properly and my end product is still turning purple.
Following